Mr. Mohd Daud
Managing Director
MOHD DAUD is an accomplished healthcare professional with over 25 years of experience in healthcare
infrastructure, operations, and technology-enabled services. He has led complex, highly regulated
healthcare environments with a strong emphasis on quality, safety, regulatory compliance, and
operational efficiency.
He brings deep expertise in end-to-end project execution, regulatory coordination, and vendor and
stakeholder management, working closely with hospitals, service providers, technology partners, and
statutory authorities. His ability to align clinical, technical, and administrative teams has been
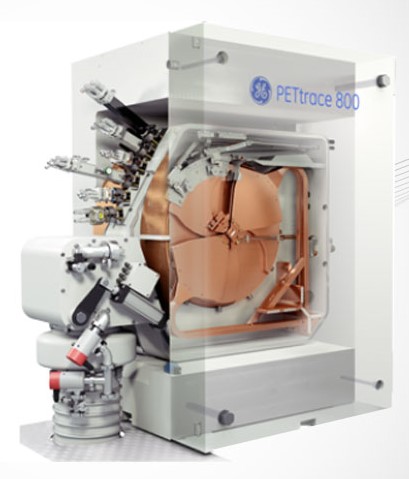
key to the successful delivery of large-scale healthcare and nuclear medicine infrastructure
projects.
As Managing Director of VRM, MOHD DAUD provides strategic direction focused on process excellence,
risk mitigation, and continuous improvement, ensuring the development of robust, scalable, and
compliant systems that support safe and efficient healthcare operations.
Read More